Aseptic Manufacturing Facilities for Injectable Drug Products
GRAM delivers aseptic fill-finish services for clinical and commercial products at our world-class facilities in Grand Rapids, Michigan, United States, featuring top-of-the-line equipment and advanced technology. Each facility adheres to cGMP standards to ensure sterility, and our talented teams implement processes with maximum efficiency to deliver exceptional results.
Three of our five GMP manufacturing sites were constructed after 2018, all with Annex 1 in mind. These facilities are highlighted below.
Sterile Filling Facility Two
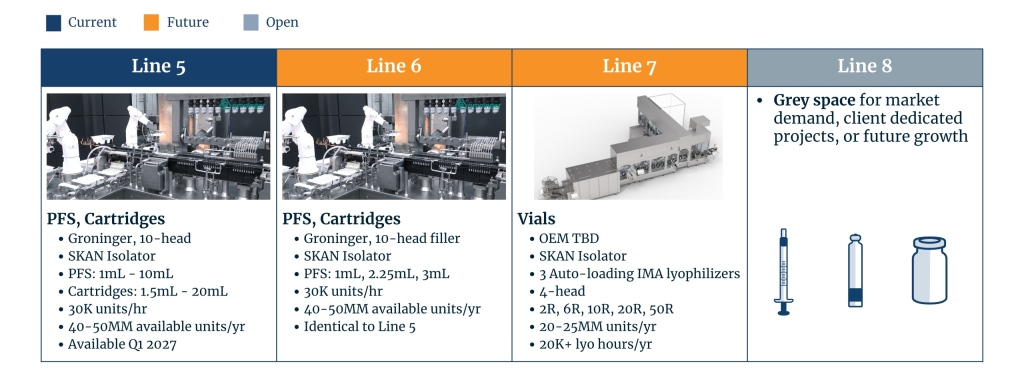
Our second sterile filling facility provides over 50 million units of syringe and cartridge filling capacity, with the groninger UFVN high-speed syringe and cartridge filling line as the starting point. At 150,000 sq ft, the facility allows for future growth and space for client-dedicated projects.

- 150,000 sq. ft.
- Grade C formulation suites
- Clean room space and utilities designed for up to 4 filling suites
- groninger 10-head syringe and cartridge filling line with SKAN isolator
- Accommodates syringe sizes ranging from 1mL to 10mL, and cartridge sizes from 1.5mL to 20mL.
- Future space and utilities to support three additional fill lines and in-line inspection (total of four fill lines)
- Viewing rooms designed for customer transparency and access
- Filled syringes are to be packaged and stored at the adjacent Finishing and Warehouse Center
Sterile Filling Facility One
Our operations are centered around a clear commitment to sterile filling. We prioritize patient safety by adhering to strict aseptic standards and collaborating closely with each client, bringing deep expertise to the partnership. Our Sterile Filling Facility One, which opened in 2020, has production lines that are approved by both the EU and the FDA. The facility spans 90,000 sq ft and has the capacity to produce over 80 million vials, and more than 10 million syringes and cartridges.

- 90,000 sq ft and three floors
- Four Grade C formulation suites and four Grade C filling rooms
- Four sterile filling lines
- Two Bausch + Ströbel 4-head Vial Fillers w/SKAN Isolators
- One Bausch + Ströbel VarioSys Vial and PFS Modular Filler w/SKAN Isolator
- One Bausch + Ströbel 8-head Vial Filler w/SKAN Isolator
- Lyophilization
- IMA LyoMax 17 Lyophilizer
- CIP/SIP Suite
- Method Development, in-process and micro laboratories
- Just-in-time warehouse
- Stand-up frozen storage -70°C
- Walk-in frozen storage -20°C
- Walk-in cold storage +2°C to +8°C
- 2021 Facility of the Year Awards Winner for Special Recognition for Operational Agility: COVID-19 Impact
Finishing and Warehouse Center

- 200,000 sq. ft.
- Terminal sterilization with two autoclaves
- Inspection:
- Three fully automated inspection systems
- Twelve semi-automated inspection systems
- Twelve manual inspection booths
- Three fully automated inspection systems
- Labeling and packaging
- Two labeling and packaging systems with broad capabilities
- Syringe labeler and assembly
- Weigh and dispense suite
- QC analytical and micro laboratory
- In-house sterility testing
- SKAN Spectra sterility isolator
- Full analytical, micro, and sterility services
- Storage
- -50°C walk-in freezers
- -50 to -80°C stand-up freezers
- -20°C, -50°C walk-in frozen storage
- +2°C to +8°C walk-in finished product
- 2-8°C and -20°C walk-in in-process combination unit
- 20-25°C controlled room temperature storage
- 50,000 sq. ft. of material and finished product storage
- Future space for auto-injector/device assembly